| A evolução humana não é somente algo do passado. Embora tenhamos mesmo evoluído durantes todos esses milhões de anos, ainda não paramos de evoluir. Isso significa que ainda há muito espaço para melhoras, e, se a civilização continuar no mesmo caminho que trilha hoje, algumas grandes mudanças podem ser esperadas para os próximos 200.000 anos. Confira dez delas: 1 – Monoetnia
Multiculturalismo é a essência da sociedade moderna. Não deve ser surpresa, então, que os seres humanos evoluam para um único grupo étnico, se a mistura das culturas continuar. Conforme a miscigenação se tornar mais comum, os seres humanos perderão lentamente as características distintivas de sua etnia, e assumirão características de diversas partes do mundo. Uma pesquisa até indicou que todos se parecerão como os brasileiros (um povo bastante miscigenado) em “pouco” tempo. Há uma vantagem óbvia nisso: “raça” já não será mais um problema. 2 – Sistema imunológico fraco
Conforme os seres humanos tornam-se mais e mais dependentes de medicamentos para a sobrevivência, o sistema imunológico vai enfraquecendo lentamente. A melhor maneira de explicar isso é com um exemplo: o uso de hormônios. Imagine um futuro em que, com a ajuda de suplementos, você possa regular seus hormônios para maximizar o seu bem-estar. Com o tempo, seu corpo se tornaria dependente dos hormônios adicionais, ao ponto de parar de fazer por si mesmo o que os suplementos podem fazer em seu lugar. Os processos que criam hormônios se tornariam menos importantes para a sobrevivência, uma vez que o seu corpo sempre tem o suficiente, graças aos suplementos. Depois de dezenas de milhares de anos, é provável que os seres humanos evoluam ao ponto de hormônios não serem mais criados organicamente dentro de nosso corpo. Se ajuda externa fosse inteiramente responsável pela nossa sobrevivência, muitas de nossas funções internas poderiam se tornar obsoletas. Por que o seu corpo precisaria de um poderoso sistema imunológico se todos os patógenos pudessem ser curados com medicação? De fato, é uma desvantagem da utilização de medicamentos para combater doenças. 3 – Menos massa muscular
Há duas causas previsíveis para o enfraquecimento físico gradual da raça humana. A primeira é a nossa crescente dependência da tecnologia – e de máquinas, em particular – para fazer o nosso trabalho sujo. Quanto menos cada geração depender da força física, mais provável é que toda a espécie fique mais fraca. A segunda causa possível para a atrofia muscular é um pouco mais impressionante: envolve um cenário em que nós temos que mudar para o espaço. Em tal cenário, a força física é quase desnecessária para o dia-a-dia. Eventualmente, perderíamos a maioria de nossa massa muscular. 4 – Mais altura
A altura humana tem crescido rapidamente nos últimos dois séculos. Ao longo dos últimos 150 anos, a altura média da espécie aumentou 10 centímetros. Acredita-se que a principal força motriz por trás deste crescimento é a abundância de nutrição disponível para muitos de nós. Quanto mais a criança tem para comer, mais energia ele ou ela tem para crescer. Enquanto tivermos a capacidade de comer em excesso, a espécie vai continuar a crescer (e ficar mais alta). Se o céu é o limite, ou se a biologia vai nos parar em algum lugar, só o tempo – e a evolução – dirá. 5 – Menos pelo
Já perdemos a maior parte do pelo do nosso corpo por uma série de razões. Seguindo esse caminho, é provável que os seres humanos se tornem ainda mais carecas ao longo do tempo. As mulheres, em particular, são frequentemente vistas como mais atraentes com menos pelo em várias partes de seus corpos. Como esse traço oferece vantagem a um indivíduo quando se trata de atratividade sexual, podemos postular que, ao longo do tempo, as mulheres evoluam para ter menos pelo. O mesmo pode ser dito para os homens, mas como há menos pressão social para que tenham pele lisa, a mudança permanente provavelmente ocorrerá mais lentamente. 6 – Mudanças cerebrais
A tecnologia já afetou a forma como a nossa memória funciona. O cérebro humano, sendo uma máquina em busca da máxima eficiência, tipicamente memoriza o ponto onde a informação é armazenada, em vez de a própria informação. É muito mais fácil de lembrar onde você colocou o livro com as informações do que recordar o conteúdo real do livro, não é mesmo? Na era da internet, essa peculiaridade mental tornou-se especialmente importante. Nós não tentamos mais decorar números de telefones, simplesmente os buscamos. Não tentamos lembrar de respostas, as pesquisamos na web,e assim por diante. Conforme a tecnologia se torna mais avançada, o nosso cérebro vai se adaptar a fim de maximizar sua eficiência, talvez em detrimento de nossa memória. 7 – Dentes menores
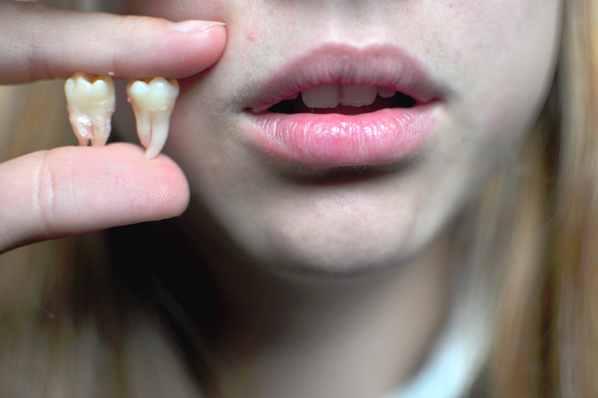
A mudança mais óbvia em nossos maxilares será o desaparecimento dos dentes do siso, que não tem mais utilidade aos seres humanos modernos. Muitos grupos étnicos já têm baixas taxas de ocorrência desse tipo de dente. Além disso, também podemos esperar que os nossos dentes fiquem menores. Ao longo da evolução do homem, tem havido uma tendência geral para dentes pequenos. Evidências mostram que nos últimos 100.000 anos, nossos dentes reduziram pela metade em tamanho. Nossos maxilares também encolheram. A tendência deve continuar, especialmente porque nossa comida é cada vez mais facilmente digerível. 8 – Menos dedos do pé
Antes dos humanos andarem eretos, nossos dedos eram usados para a luta, assim como nossas mãos. Conforme dependemos menos da escalada e mais de ficar de pé, nossos pés têm lentamente se reduziram ao seu tamanho atual. A evolução agora caminha para livrar-nos do nosso quinto dedo do pé, o menor. Em comparação com os dedos maiores que servem para nos dar equilíbrio e andar, os pequenos não servem de nada, e podemos sobreviver muito bem sem eles. Devido a isso, e por causa dos problemas que surgem a partir de sua existência desnecessária – como serem frequentemente esmagados em sapatos e em esbarrões com objetos -, podemos esperar que os humanos se tornem uma criatura de quatro dedos. 9 – Crânios menores ou maiores
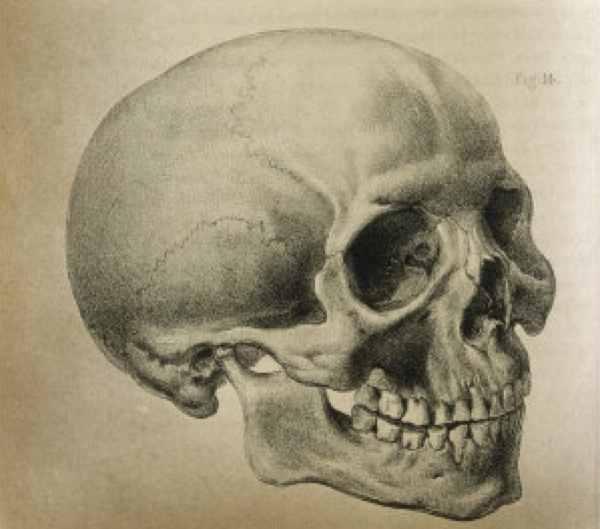
Duas escolas de pensamento existem sobre a questão do volume do nosso crânio. Uma, que conta com o apoio de muitos cientistas, afirma que nosso crânio está no limite de seu tamanho. Qualquer pessoa que tenha dado à luz sabe que a cabeça de uma criança já é, para falar diplomaticamente, bastante grande. Por esta razão, muitos biólogos acreditam que uma cabeça maior tornaria o nascimento impossível – algo que o processo evolutivo eliminaria gradualmente rapidamente, sem dúvida. A grande cabeça no nascimento é também mais propensa a ferir ou matar a mãe. Assim, parece inevitável que o tamanho da nossa cabeça fique o mesmo, ou até menor. No entanto, isso ignora o fato de que cesarianas são comuns e oferecem oportunidades para a sobrevivência de crianças com grandes cabeças. Na verdade, alguns acreditam que a cesárea acabará por ser mais segura do que o parto natural no futuro, o que leva à possibilidade de que as crianças com cabeças pequenas, naturalmente entregues, sobrevivam menos. Mas tal dependência seria perigosa para os seres humanos. Se humanos “cabeçudos” perdessem a capacidade de realizar cesarianas, poderíamos esperar uma extinção rápida. 10 – Autoevolução
Os seres humanos podem, eventualmente, chegar a um ponto no qual “forcem” a evolução em si mesmos através do uso da tecnologia. Seja através de órgãos biônicos, por exemplo, ou por meio de seleção genética, na qual futuros pais escolhem as características de seu filho antes do nascimento, a evolução humana deverá caminhar por essa estrada. A seleção genética, em particular, pode levar rapidamente a um boom de “bebês projetados”, nos quais todos os defeitos e traços indesejáveis podem ser removidos. Se isso se generalizar, poderia potencialmente forçar muitos traços humanos (negativos ou não) à extinção.[Listverse] http://hypescience.com/10-possiveis-proximos-passos-da-evolucao-humana/
|